Admissions Period
Now accepting applications for B.S. Biomanufacturing until July 31, 2026.
Bachelor of Science in Biomanufacturing
The Biomanufacturing Bachelor’s degree program develops students' skills, abilities, and knowledge to work in the unique biological manufacturing environment. The full-time program prepares students for employment in technical or quality positions in the manufacturing sector of the biotechnology industry, which includes biotherapeutics, diagnostics, supplies and services, and industrial products.
Why Biomanufacturing?
A growing regional industry.
Los Angeles County is home to nearly 2,900 bioscience companies and research institutions, while Ventura County supports more than 8,000 workers in life sciences through major employers in Thousand Oaks, Camarillo, and Simi Valley. Santa Barbara County adds to this regional network with companies and institutes engaged in biomedical, agricultural, and marine sciences.
Strong career outlook.
Biomanufacturing professionals are in high demand across the region. Salaries generally range from $40,000 to $97,000, with projected job growth of approximately 20% over the next five years.
Preparation for leadership.
Our program includes supervisory-level coursework in quality assurance, project management, ethics, and government regulation, equipping graduates with the skills needed to advance into leadership positions in the life science sector.
Pathways across industries.
Skills developed through biomanufacturing are applicable in pharmaceutical, medical, agricultural, chemical, manufacturing, mechanical, and engineering fields, providing graduates with flexibility in career direction.
Hands-on experience with industry partners.
Through established partnerships, students have opportunities for internships and apprenticeships with local life science companies, gaining practical experience and direct pathways to employment.
The B.S. in Biomanufacturing program launched Fall 2024 at Moorpark College.
Who should complete the Program Interest Form?
Prospective Students, K-12 Partners, Community Members are invited to submit an interest form and/or questions about the bachelor degree program at Moorpark College. You can also email MCbdp@vcccd.edu
Prospective students
We encourage you to meet with a counselor to assess your readiness for the program and to consider applying in a future admissions cycle.
Program Requirements, Curriculum, and Course Outlines
The Bachelor of Science in Biomanufacturing is 120 units which includes:
Prerequisite Lower-Division & General Education Courses Units*
CHEM M01A/M01AH: General Chemistry I
C-ID: CHEM 110, CHEM 120S (with CHEM M01B)
Studies atomic theory and stoichiometry; nomenclature and chemical reactions; thermochemistry; quantum theory and the electronic structure of atoms; chemical bonding and molecular structure; physical behavior of gases; states of matter and phase equilibria; and solutions. Addresses, through hands-on laboratory activities, spectroscopy; distillations; quantitative, qualitative and statistical analyses; titrations; thermochemistry; gravimetric and volumetric analyses; and colligative properties.
5 units
CHEM M01B: General Chemistry II
C-ID: CHEM 120S (with CHEM M01A or CHEM M01AH)
Examines chemical kinetics; phase equilibria; equilibria in gases and solutions; acids and bases; solubility and complex ions; thermodynamics; electrochemistry; qualitative and quantitative chemical analyses. Provides an overview of nuclear chemistry, coordination chemistry, and organic chemistry. Addresses, through hands-on laboratory activities, chemical kinetics; equilibria; thermodynamics; spontaneous oxidation-reduction reactions and electrolysis; selective precipitation; titrations; and exposure to ultraviolet, infrared, and nuclear magnetic resonance spectroscopy.
5 units
BIOL M02A/M02AH: General Biology I/Honors
C-ID: BIOL 190, BIOL 135S (with BIOL M02B)
Introduces students to major biological themes and principles that are fundamental to an understanding of life processes in any field of biology today. Includes the scientific process, experimental design, biological chemistry, prokaryotic and eukaryotic cell structure and function, cellular metabolism, cell reproduction and its controls, cell communication, genetics, molecular biology, DNA technology and evolutionary processes. Credit Limitation: Credit will not be awarded for both the honors and regular versions of a course. Credit will be awarded only for the first course completed with a grade of "C" or better or "P". Moorpark College Honors program requires a letter grade.
5 units
MICR M01: General Microbiology
C-ID: N/A
Emphasizes microbiological principles and lab techniques related to the morphology, metabolism, genetics, classification, and ecology of bacteria. Characterizes viruses and eukaryotic microorganisms. Focuses on human disease including characteristics of pathogens, immunology, and epidemiology. Practices laboratory exercises including aseptic technique, staining, the determination of bacterial growth conditions and requirements, and the identification of an unknown bacterium. Applies microbiology in clinical applications.
5 units
BIOT M02A/BIOL M12A: Environmental Control and Process Support
C-ID: N/A
Provides skills training in manufacturing of biopharmaceuticals and medical devices. Presents an overview of the manufacturing process and introduces environmental control and process support with a focus on Good Laboratory Practices (GLP)/Good Manufacturing Practices (GMP), clean room procedure, monitoring techniques, and required documentation.
2 units
BIOT M02B/BIOL M12B: Manufacturing: Quality Control and Validation
C-ID: BIOT 210X
Provides skills training in industrial biotechnology with emphasis on manufacturing of pharmaceuticals and medical devices. Introduces validation and quality control. Reviews manufacturing process, including formulation, lyophilization, packaging and filling. Focuses on validation, systems evaluations, testing and reporting.
2 units
BIOT M02C/BIOL M12C: Manufacturing: Cell Culture and Microbial Fermentation (laboratory)
C-ID: BIOT 230X
Provides skills training in industrial biotechnology with emphasis on manufacturing pharmaceuticals. Introduces cell culture and microbial fermentation. Focuses on bacterial techniques, microbial assessment, mammalian cell culture, bioreactor fermentation, and media preparation. Compares small and large industrial scale cell culture.
3 units
BIOT M02D/BIOL M12D: Bioprocessing: Recovery and Purification
C-ID: BIOT 220BX
Provides skills training in industrial biotechnology with emphasis on manufacturing pharmaceuticals. Introduces bioprocessing, recovery, and purification techniques. Focuses on protein separation and purification, chromatography, large-scale recovery, and identification of assays. Reviews skills necessary for a successful job search in the field of biotechnology.
2 units
BIOT M02E/BIOL M12E: Business Practices and Governmental Regulation
C-ID: N/A
Provides skills training in industrial biotechnology with emphasis on manufacturing pharmaceuticals. Examines manufacturing from the perspective of company operations involved with the drug or medical device development process. Focuses on business practices and governmental regulations.
2 units
BIOT M10/BIOL M13: Introduction to Biotechnology and Molecular Biology (laboratory)
C-ID: BIOT 150BX
Examines the role of molecular biology in the manufacturing of commercial pharmaceutical and agricultural products. Introduces basic biotechnology laboratory skills, including documentation, safety, and solution and buffer preparation. Develops student proficiency in aseptic techniques, spectrophotometry, molecular biology techniques, and electrophoresis.
4 units
ENGL M01A/M01AH: English Composition
C-ID: ENGL 100
Emphasizes expository writing and research, demonstrating principles of thesis and support, rhetorical organization, control of diction, clear sentence and paragraph construction, and command of the conventions of English usage. Develops analytical and interpretive reading skills.
4 units
MATH M15/M15H: Introductory Statistics/Honors
C-ID: MATH 110
Explores the nature of statistical methods, including description of sample data, probability, theoretical frequency distributions, sampling, estimation, testing hypotheses and special topics. Provides problem-solving techniques.
4 units
Upper-Division Major Courses
BIOT M301: Biomanufacturing Process Sciences (lecture/lab)
Physical and chemical principles of biochemical engineering that enable large cell culture-Thermodynamics and the properties of fluids; mass and heat transfer, fluid flow, and the energy relationships in fluid systems-Biomanufacturing technologies enabling large-scale upstream and downstream processes
5 Units
BIOT M302: Design of Experiments for Biomanufacturing(lecture/lab)
Established methods to systematically vary process parameters to improve and optimize a biomanufacturing process
4 Units
BIOT M303: Design of Biomanufacturing Facilities, Critical Utilities, Processes, and Equipment (lecture)
An examination of how the robust design of all aspects of a biomanufacturing facility minimizes errors-The role of Quality by Design (ICH Q8) in facility design-Processes and equipment in biological production, recovery, and purification-Aseptic process design-Clean utility and support systems
4 Units
BIOT M304: Bioprocess Monitoring and Control (lecture/lab)
The measurement, monitoring, modeling, and control of biomanufacturing processes
5 Units
BIOT M307: Advanced Topics in Quality Assurance and Regulatory Affairs (lecture)
Study of the harmonized quality system approaches of ICH Q8, 9, 10, and 11, including quality risk management, qualification, and validation
4 Units
BIOT M405: Cell and Gene Therapy Manufacturing Technologies (seminar)
An examination of the emerging field of cell and gene therapy manufacturing for cancer therapy
3 Units
BIOT M406: Supply Chain and Enterprise Resource Planning (lecture)
Manage flow of materials in a supply chain-Understand the design, planning, and execution of raw material procurement and use-Eligibility for certification test
3 Units
BIOT M408: Six Sigma and Lean Manufacturing (lecture/discussion)
Study of key six sigma concepts and tools; the DMAIC phases: design, measure, analyze, improve, and control-Use and implementation of lean tools to reduce waste -Completion of this course prepares students to earn a certification in six sigma
4 Units
BIOT M409: Methods in Quality Improvements, Investigations, and Audits (lecture)
The study of continuous quality improvement techniques, including investigational methods into process deviations
4 Units
BIOT M410: Emerging Trends in Biomanufacturing Quality (seminar)
An examination of new regulatory requirements and changes to current practices in biomanufacturing quality
3 Units
Total Upper Division Major Course Units: 39
Upper Division General Education Courses
ENGL M300: Technical Writing
Concentrated work in technical writing focused on communicating the results of technical work at a high level of competence
3 Units
BUS M400: Project Management
Core characteristics of project management including planning, execution, monitoring, and control
3 Units
PHIL M400: Ethics in the Age of Emerging Technology
Application of ethical theories to contemporary ethics issues and problems
3 Units
Total Upper Division General Education Units: 9
Link to Course Outlines: https://catalog.vcccd.edu/course-search/
Takeda sponsored the Analytical Lab to provide our biotechnology students with hands-on experiences, equipping them with the analytical and technical skills essential for success in the biotech manufacturing industry. The lab directly supports our students to gain the analytical and technical skills needed to thrive in the industry through hands-on experiences.

Now accepting applications for B.S. Biomanufacturing until July 31, 2026.
Applying to the California Community College Bachelor's Degree program is a two-step process.
All Bachelor Degree Program (BDP) Applicants must first apply for Admission to Moorpark College via the California Community College CCCApply. Becoming a student at Moorpark College is free and easy.
Moorpark College/VCCCD students receive:
• Ventura County Community College District (VCCCD) Student ID 900#
• myVCCCD Email Address
• Access to Bachelor Degree Program (BDP) Admissions Application for the B.S. in Biomanufacturing program
Note for Alumni/Returning Students:
• Welcome back! All students, current, returning, and new, must complete a CCCApply application and select a BS program to gain access to the BS online admissions application
• Returning students may make changes to their e-mail address, phone number, or address on the MyVCCCD Student Portal
• If your name has changed since you last attended, please visit the Admissions & Records Office to make the change.
CCCApply Guidance & Screenshots
The Bachelor's Degree Program Application will be sent to prospective students as part of the Moorpark College Acceptance Letter.
Step 1. Apply/Re-Apply to Moorpark College via CCCApply
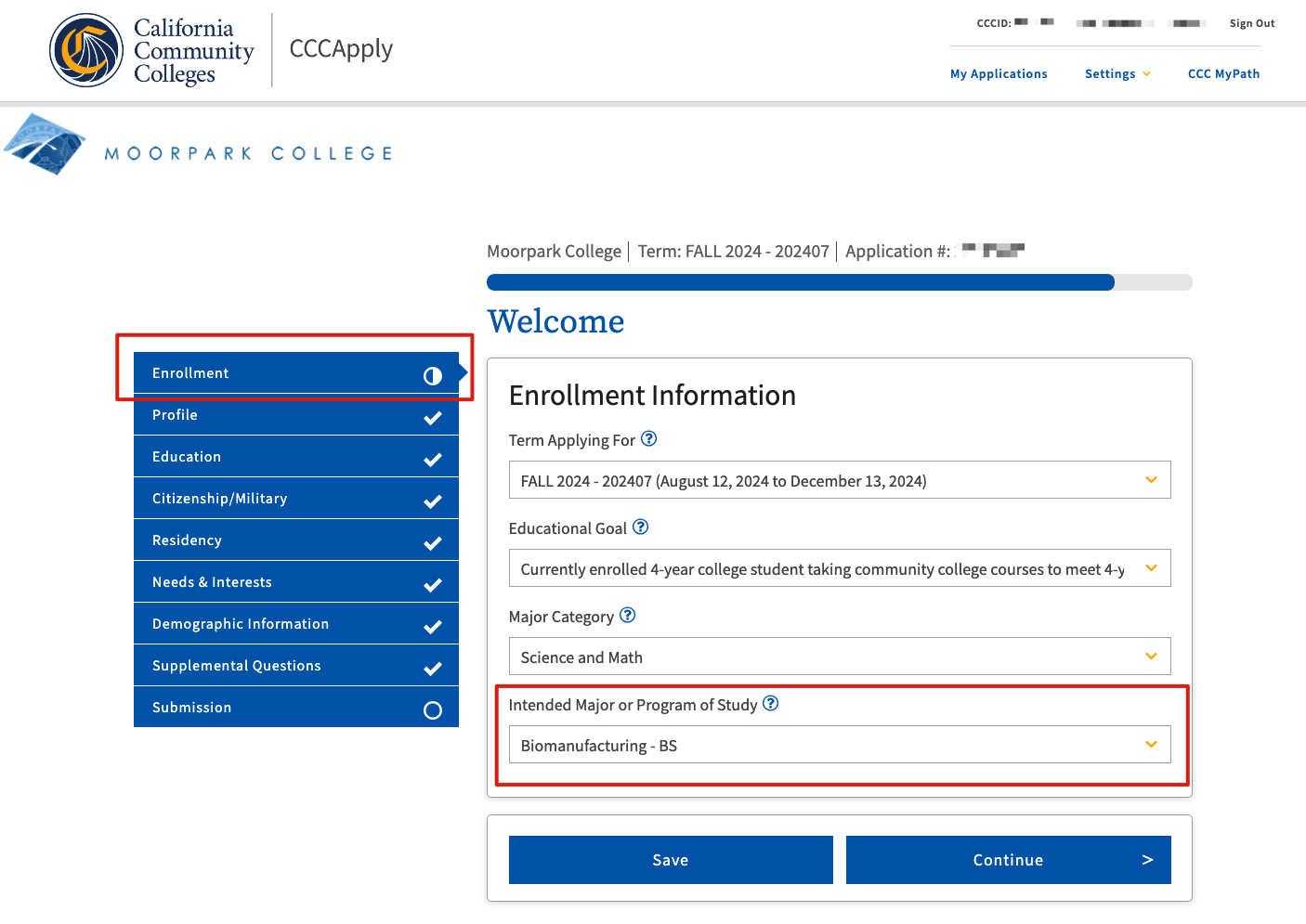
Step 2. Indicate the Term, Educational Goal, Major Category, and BDP Program you are applying to. Example below: Fall 2024, 4-year college student, Science & Math, BS Biomanufacturing (available February 1 - July 31, 2024)
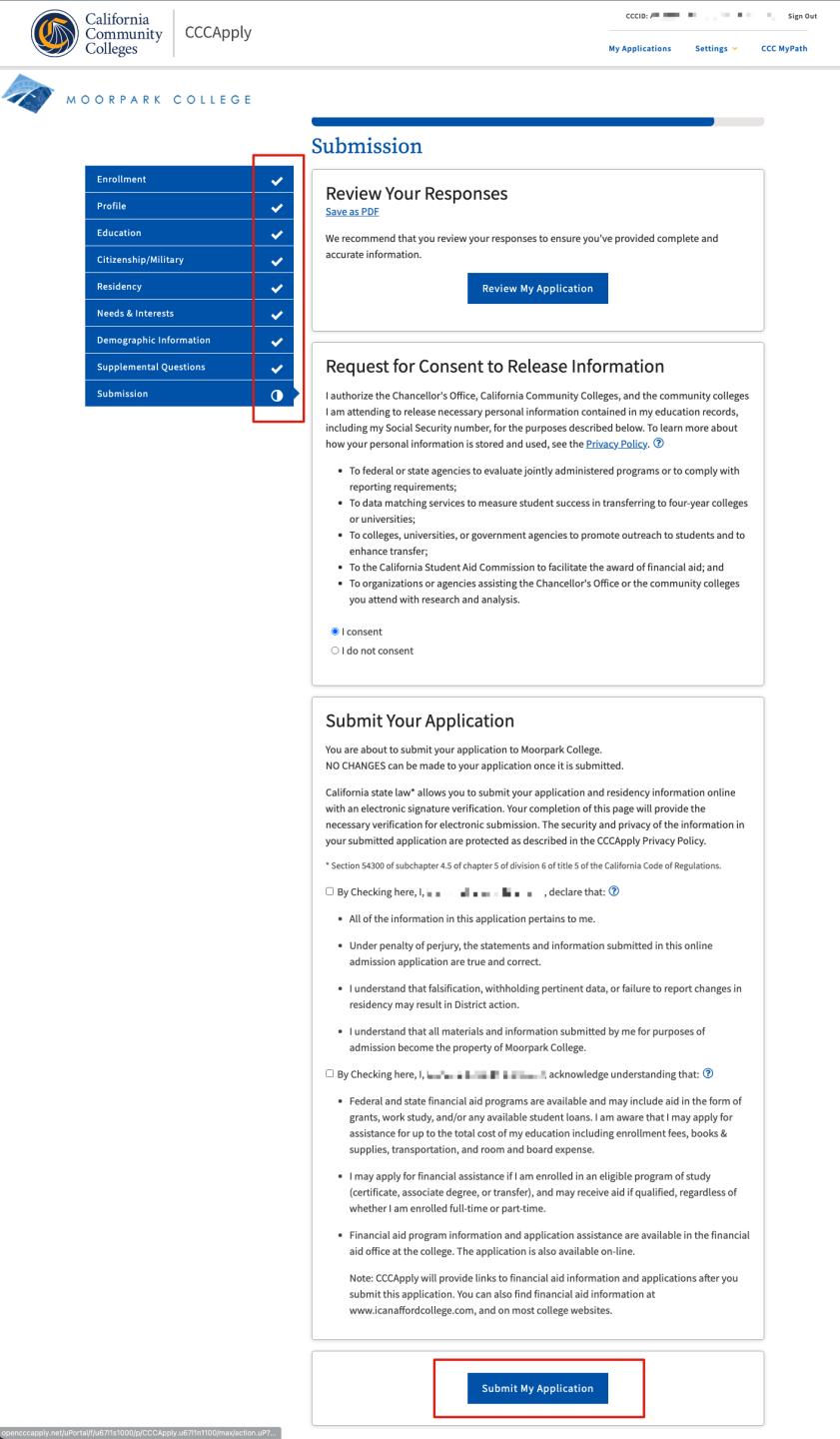
Step 3. Complete all remaining sections of the application: Profile, Education, Citizenship/Military, Residency, Needs & Interests, Demographic Information, Supplemental Questions, and Submit Application
Step 4. Save a copy of your Application
Step 5. Check your emails regularly for Admissions Acceptance Letter, including your 900#, myVCCCD email address, and link to the Online Bachelor Degree Program Application.
Questions about the Moorpark College Acceptance process can be directed to 805-378-1429 or mcadmissions@vcccd.edu.
The B.S. in Biomanufacturing builds on the Biotechnology program, and the required prerequisites are met with:
• AS in Biotechnology OR
• 60 units in college credit, including the Certificate in Biotechnology, Freshman Composition, and Statistics, with a GPA of 2.0.
Students exploring the biotechnology field, and/or interested in learning more about the program are encouraged to complete the Program Interest Form.
Students and community partners who submit an interest form will be notified of application orientations, application dates, and other program information as it becomes available.
High School Articulation for Biotechnology
The following local high schools have articulation agreements with Moorpark College. Students who completed one of the articulated courses below may receive equivalent credit for BIOT M01A, a pre-requisite B.S. in Biomanufacturing course.
Foothill Technology High School
| MC Class # | MC Class Title | No. of Units | CSU/UC Transferable | HS Class Title |
| BIOT M01A | Intro to Biotechnology & Molecular Biology I | 4 | CSU/UC | Introduction to Biotechnology |
Newbury Park High School
| MC Class # | MC Class Title | No. of Units | CSU/UC Transferable | HS Class Title |
| BIOT M01A | Intro to Biotechnology & Molecular Biology I | 4 | CSU/UC | Biotechnology Research & Development CP |
Simi Valley High School
| MC Class # | MC Class Title | No. of Units | CSU/UC Transferable | HS Class Title |
| BIOT M01A | Intro to Biotechnology & Molecular Biology I | 4 | CSU/UC | Biotechnology Research & Development CP |
Thousand Oaks High School
| MC Class # | MC Class Title | No. of Units | CSU/UC Transferable | HS Class Title |
| BIOT M01A | Intro to Biotechnology & Molecular Biology I | 4 | CSU/UC | Scientific Research |
Westlake High School
| MC Class # | MC Class Title | No. of Units | CSU/UC Transferable | HS Class Title |
| BIOT M01A | Intro to Biotechnology & Molecular Biology I | 3 | CSU/UC | Introduction to Biotechnology |
To view a full list of all existing High School Articulation courses visit: https://www.moorparkcollege.edu/career-pathways/articulation
When did the bachelor degree program begin?
Fall 2024
When can I apply?
Students are encouraged to apply the Spring prior to completing all required prerequisites.
What courses do I need to apply?
Students apply as Juniors (60 units of college credit). All lower division courses listed as "prerequisites" must be completed or in progress to be completed before admission.
See the complete list of prerequisites under "Curriculum & Courses".
Is it required that all lower-division courses be completed at Moorpark College?
Lower division courses in subjects like BIOL, CHEM, MATH, and ENGL can generally be completed at most California Community Colleges. You can verify equivalency by consulting your counselor or checking the C-ID in the catalog. However, since biotechnology courses may not have equivalents at other community colleges, some must be taken at Moorpark College or at a community college where a C-ID is available. For specific course inquiries, please consult your counselor to determine your readiness for the program.
What is the program schedule?
The B.S. in Biomanufacturing is a bachelor degree completion program is a cohort model. Students will attend 2 full-days per week. Fall 2025 students will attend Monday & Wednesday. Fall 2026 is anticipated to attend Tuesday & Thursday.
Can I attend part-time?
The B.S. in Biomanufacturing is a bachelor degree completion program designed as a cohort model. Students who are accepted to the program are expected to enroll in the full-time program for a total of 4 semesters.
How much will the program cost?
The State of California sets the tuition for the Community College Bachelor Degree Program at a maximum of $130 per unit. Students who attend may also apply for financial aid and scholarships towards tuition.
Apply for Financial Aid, Moorpark College School Code: 007115:
https://studentaid.gov/h/apply-for-aid/fafsa
Did we miss your question? Visit the Bachelor Degree Program Website FAQ or contact us at MCbdp@vcccd.edu